Why I Will Never Recommend Ozempic: A Personal Story and Clinical Reality Check
I watched my dad go through it. The gallbladder problems. The neuropathy. The GI issues that never fully resolved. All from a medication his doctor prescribed to help manage his diabetes.
He trusted the system. He took the medication as directed. And he paid for it with his health.
This is not an anti-medicine rant. I am a peptide researcher who believes in pharmacological intervention when it makes sense. But watching someone I love suffer through preventable complications changed how I think about semaglutide, and I am going to tell you exactly why Ozempic should not be the default prescription for diabetics.
KEY FACTS
Ozempic face is real: 25-40% of weight lost on semaglutide comes from lean mass (muscle), causing facial hollowing, sagging skin, and premature aging.
Over 2,000 lawsuits have been filed against Novo Nordisk for gastroparesis, vision loss, and other severe complications as of early 2026.
Gallbladder disease was added to the warning label in March 2022 after widespread reports.
Gastroparesis (stomach paralysis) affects approximately 1% of users, with some cases persisting for over a year after stopping the drug.
Vision loss from NAION is now a documented risk, with European regulators adding it to product warnings in June 2025.
35% of weight lost is lean mass, not fat, potentially worsening long-term metabolic health.
What Happened to My Dad
My dad was prescribed Ozempic for type 2 diabetes. His doctor told him it would help with blood sugar control and he might lose some weight as a bonus. Standard pitch. Standard prescription.
Within months, the problems started.
The gallbladder came first. Severe abdominal pain that landed him in the emergency room. Turns out the rapid metabolic changes from semaglutide can trigger gallbladder disease. He ended up needing his gallbladder removed. The FDA added gallbladder disease to the Ozempic warning label in March 2022, but his doctor never mentioned it as a risk.
Then came the neuropathy. Tingling and numbness in his extremities that his doctors initially attributed to his diabetes itself. But the timing lined up with semaglutide use, and the symptoms worsened on the medication rather than improved. Diabetic neuropathy should stabilize or improve with better glucose control. His got worse.
The GI issues never stopped. Constant nausea. Irregular bowel movements. Feeling full after two bites of food. His quality of life tanked. He was technically "healthier" by the numbers on paper, but he felt worse every day.
He eventually stopped the medication. Some symptoms improved. Some did not. The neuropathy persists. The gallbladder is gone forever.
This is not a rare story. Over 2,000 people have filed lawsuits against Novo Nordisk for similar experiences. The FDA has updated the Ozempic label multiple times since 2022 to add warnings the company should have disclosed from the start.
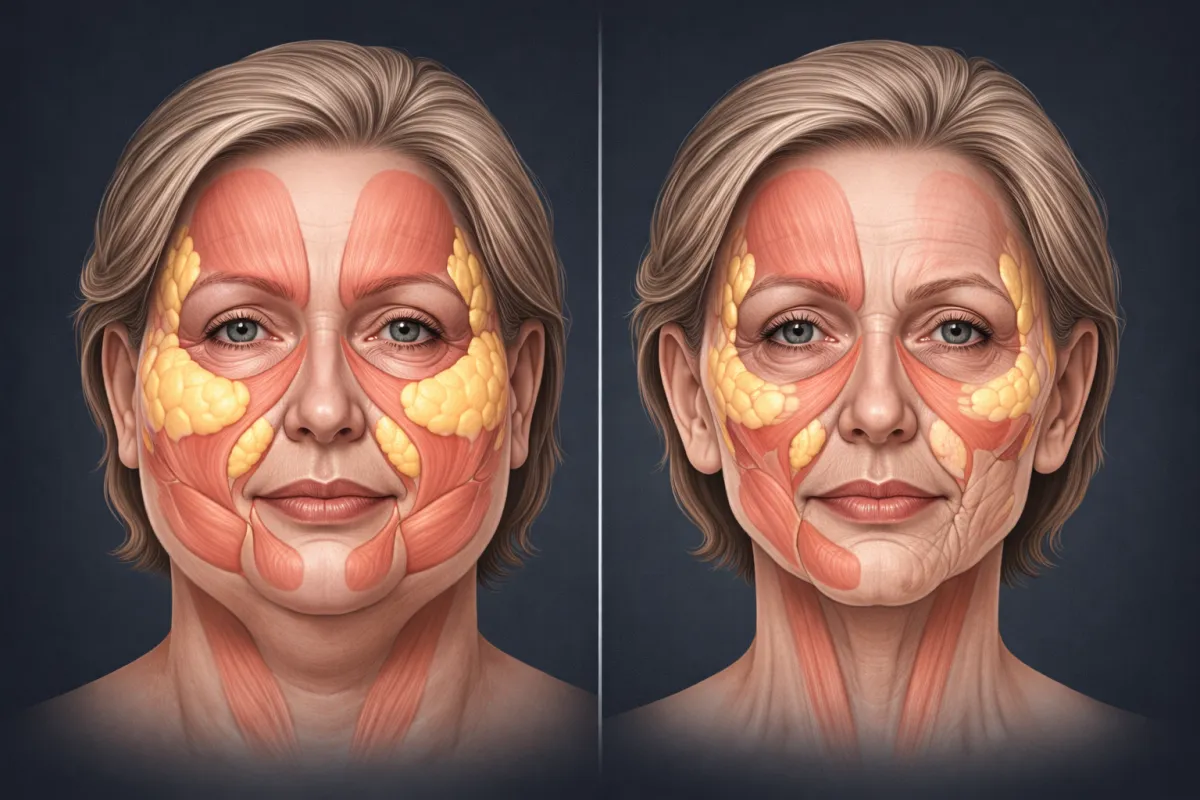
Ozempic Face: The Problem Everyone Sees But Nobody Talks About
Scroll through any celebrity news site and you will see it: sunken cheeks, hollow eyes, sagging jowls on people who used to look vibrant and healthy. They call it "Ozempic face."
The term was coined by dermatologist Dr. Paul Jarrod Frank after treating wave after wave of patients with the same pattern: rapid weight loss followed by facial aging that made them look ten to fifteen years older than before.
Here is what is actually happening:
You are not just losing fat. You are losing muscle.
Research shows that 25-40% of weight lost on GLP-1 medications like Ozempic comes from lean mass, not fat. For every 10 pounds you lose, 2.5 to 4 pounds is muscle, organ tissue, and structural support.
Your face depends on subcutaneous fat and underlying muscle to maintain its shape. When you strip both away rapidly, the skin has nothing to hold onto. It sags. It wrinkles. It hollows out.
This is not just cosmetic. It is metabolically dangerous.
Muscle is your primary driver of glucose metabolism. It is where insulin sensitivity lives. Losing muscle mass makes diabetes harder to control in the long run, not easier. You might see better numbers in the short term because you are eating less, but you are degrading the very tissue that processes glucose.
The cruel irony: a diabetes medication that destroys the tissue most responsible for blood sugar regulation.
Women and older adults lose the most muscle.
A 2025 study from the Endocrine Society found that women and older adults on semaglutide lost disproportionately more muscle than younger men. The people most likely to be prescribed this medication are often the ones most vulnerable to its muscle-wasting effects.
The Side Effects They Did Not Tell You About
Novo Nordisk made over $25 billion in worldwide semaglutide sales in 2024. That is a lot of incentive to downplay problems.
Here is what the warning label did not adequately disclose until lawsuits and FDA pressure forced changes:
Gastroparesis (Stomach Paralysis)
Approximately 1% of Ozempic users develop gastroparesis, a condition where the stomach cannot empty properly. Symptoms include severe nausea, vomiting, bloating, and abdominal pain. Some cases require hospitalization. Some require surgery.
The mechanism makes sense: GLP-1 agonists work partly by slowing gastric emptying. In some people, this effect becomes pathological. The stomach essentially stops functioning.
What the lawsuits allege: Novo Nordisk knew about this risk but deliberately avoided listing "gastroparesis" on the warning label. They used softer language about "delayed gastric emptying" instead. The FDA finally updated the label in January 2025 to say Ozempic is "not recommended in patients with severe gastroparesis." Note: it still does not say Ozempic causes gastroparesis.
Some patients report gastroparesis symptoms persisting for over a year after stopping the medication.
Vision Loss (NAION)
Non-arteritic anterior ischemic optic neuropathy (NAION) is a condition where blood flow to the optic nerve is disrupted, causing sudden, often permanent vision loss.
A 2024 study found that semaglutide users had significantly elevated risk of NAION:
Diabetic patients: 4x increased risk
Weight loss patients: 7x increased risk
In June 2025, European regulators added NAION to the Ozempic label as a "very rare" side effect. Vision loss lawsuits are now being filed in multiple states.
Gallbladder Disease
Rapid weight loss triggers gallstone formation. Semaglutide causes rapid weight loss. The connection is not complicated.
Gallbladder disease was added to the warning label in March 2022. Many patients, like my dad, were prescribed the medication before this warning existed or without being informed of the risk.
Pancreatitis
Acute pancreatitis, including fatal cases, has been reported with semaglutide. The January 2025 label update explicitly states:
"Acute pancreatitis, including fatal and non-fatal hemorrhagic or necrotizing pancreatitis, has been observed in patients treated with GLP-1 receptor agonists, including semaglutide."
Acute Kidney Injury
Dehydration from vomiting and diarrhea can lead to kidney damage. Multiple cases have been reported to the FDA.
Intestinal Blockage
Ileus (intestinal obstruction) was added to the warning label in September 2023 after reports of severe cases requiring surgical intervention.
The Muscle Loss Problem Nobody Wants to Address
This is the issue that bothers me most because it undermines the entire premise of the medication.
The COURAGE trial confirmed it: 35% of semaglutide-induced weight loss is lean mass, not fat.
Think about what that means for a diabetic patient:
Muscle is the primary tissue for glucose disposal
More muscle means better insulin sensitivity
Less muscle means worse long-term glucose control
Semaglutide destroys muscle while "treating" diabetes
You are treating the symptom (high blood sugar) while worsening the underlying problem (insufficient muscle mass for glucose metabolism).
Pharmaceutical companies are now racing to develop add-on drugs that preserve muscle during GLP-1 therapy. The BELIEVE trial showed that combining semaglutide with bimagrumab (a muscle-preserving drug) helped protect lean mass. Regeneron's COURAGE trial tested similar combinations.
Why are we prescribing a medication that requires a second medication to fix its damage?
Why Diabetics Specifically Should Be Cautious
The standard argument goes: "Obesity causes diabetes. Ozempic causes weight loss. Therefore Ozempic helps diabetes."
This is too simple.
Not all weight loss is equal.
Losing 50 pounds of fat while preserving muscle would genuinely improve diabetic outcomes. Losing 50 pounds of mixed fat and muscle might improve short-term numbers while setting up long-term metabolic decline.
Diabetics already have compromised muscle metabolism.
Diabetes is associated with accelerated muscle loss (sarcopenia) and reduced muscle quality. Adding a medication that further degrades muscle tissue is throwing gasoline on a fire.
Gastroparesis is already common in diabetics.
Diabetic gastroparesis affects roughly 5% of type 1 diabetics and 1% of type 2 diabetics over a decade. Adding a medication that causes additional gastric slowing to a population already at risk for gastroparesis is questionable judgment.
The neuropathy connection needs more investigation.
My dad's experience is not isolated. There are reports of worsening neuropathy in diabetic patients on semaglutide. Whether this is direct causation or correlation with rapid metabolic shifts needs research that, conveniently, has not been funded.
What I Would Tell My Dad Now
If I could go back in time, here is what I would tell him:
First, try everything else.
Resistance training increases muscle mass, which directly improves insulin sensitivity. Dietary changes that emphasize protein and reduce processed carbohydrates address the root cause. Sleep optimization, stress management, and movement throughout the day all contribute to glucose regulation.
These interventions do not make pharmaceutical companies billions of dollars, so they are not marketed as aggressively. But they work, and they do not cause gallbladder disease.
If medication is necessary, understand the full risk profile.
Know about gastroparesis. Know about gallbladder disease. Know about vision loss. Know about muscle wasting. Make an informed decision with all the data, not just the marketing materials.
Consider alternatives to single-target GLP-1 agonists.
If incretin therapy makes sense, triple agonists like retatrutide add glucagon receptor activation, which increases energy expenditure and may have better body composition outcomes. The glucagon component theoretically promotes fat burning over muscle catabolism.
This is still being studied. But at minimum, the next generation of these drugs was designed with awareness of the muscle loss problem that first-generation drugs like semaglutide ignore.
Monitor aggressively.
If you do use semaglutide, track more than just blood sugar and weight. Get DEXA scans to monitor body composition. Watch for early signs of gastroparesis. Report vision changes immediately. Do not assume side effects are "normal" just because they are common.
The Honest Conclusion
Ozempic works for weight loss. The clinical trials prove it. People lose significant amounts of weight.
But "works for weight loss" is not the same as "improves health outcomes." Especially when:
35% of that weight loss is muscle
1% of users develop stomach paralysis
Vision loss is a documented risk
Gallbladder removal is common enough to warrant a label warning
Over 2,000 lawsuits have been filed for undisclosed side effects
I watched my dad suffer through complications that were predictable, preventable, and inadequately disclosed. His doctors trusted the pharmaceutical company's messaging. He trusted his doctors. The system failed him.
I am not telling you never to use semaglutide. I am telling you to go in with eyes open, demand full disclosure of risks, and consider whether the trade-offs make sense for your specific situation.
For diabetics specifically, I believe the muscle-wasting effects make Ozempic a poor first-line choice. You are treating blood sugar while destroying the tissue most responsible for processing blood sugar. That math does not add up.
There are better options emerging. There are lifestyle interventions that address root causes. There are combination approaches that might preserve muscle while achieving metabolic benefits.
But those do not generate $25 billion in annual revenue, so you will not hear about them from the same sources pushing Ozempic.
Research Citations
Gastroparesis and GI Complications: Chaudhry A, et al. "Tendency of Semaglutide to Induce Gastroparesis: A Case Report." PMC. 2024. Documented complete resolution of gastroparesis symptoms after semaglutide discontinuation.
NAION Vision Loss: Hathaway JT, et al. "Risk of nonarteritic anterior ischemic optic neuropathy in patients prescribed semaglutide." JAMA Ophthalmology. 2024. Found 4x increased risk in diabetics and 7x increased risk in weight loss patients.
Muscle Loss Data: COURAGE Trial Interim Results. Regeneron Pharmaceuticals. September 2025. Confirmed 34.5% of semaglutide-induced weight loss was lean mass.
Lean Mass Preservation Research: "Preservation of lean soft tissue during weight loss induced by GLP-1 and GLP-1/GIP receptor agonists: A case series." SAGE Open Medicine. 2025. Documented 26-40% lean mass loss in GLP-1 therapy across multiple trials.
Ozempic Face Dermatological Research: Montecinos, et al. "Semaglutide 'Ozempic' Face and Implications in Cosmetic Dermatology." Dermatological Reviews. 2024. Described the mechanism of facial volume loss and premature aging.
FDA Label Updates:
March 2022: Gallbladder disease added
September 2023: Ileus and intestinal blockages added
November 2024: Pulmonary aspiration during anesthesia added
January 2025: Severe pancreatitis and kidney injury warnings updated; gastroparesis contraindication added
Lawsuit Documentation: As of February 2026, over 2,190 cases consolidated in MDL before Judge Karen S. Marston, Eastern District of Pennsylvania.
Disclaimer: This content is for educational purposes only and reflects personal experience and publicly available research. This is not medical advice. Consult qualified healthcare professionals before making medication decisions. If you are currently taking Ozempic, do not discontinue without medical supervision.